All spontaneous chemical processes release energy. However, many of the processes at the basis of Life are non-spontaneous, thus require the absorption of energy, rather than its loss into heat. Therefore, an important question is: can we understand and engineer the molecular mechanisms allowing energy-demanding processes? In principle, addressing this question would enable us to perform any chemical reaction, regardless of its energetic requirements. This is the reason why ours is the Laboratory of Driven Chemical Processes.
Processes enabled by chemical driving include energy storage, active transport phenomena, the formation of seemingly impossible materials, the emergence of oscillations and patterns and memory effects.
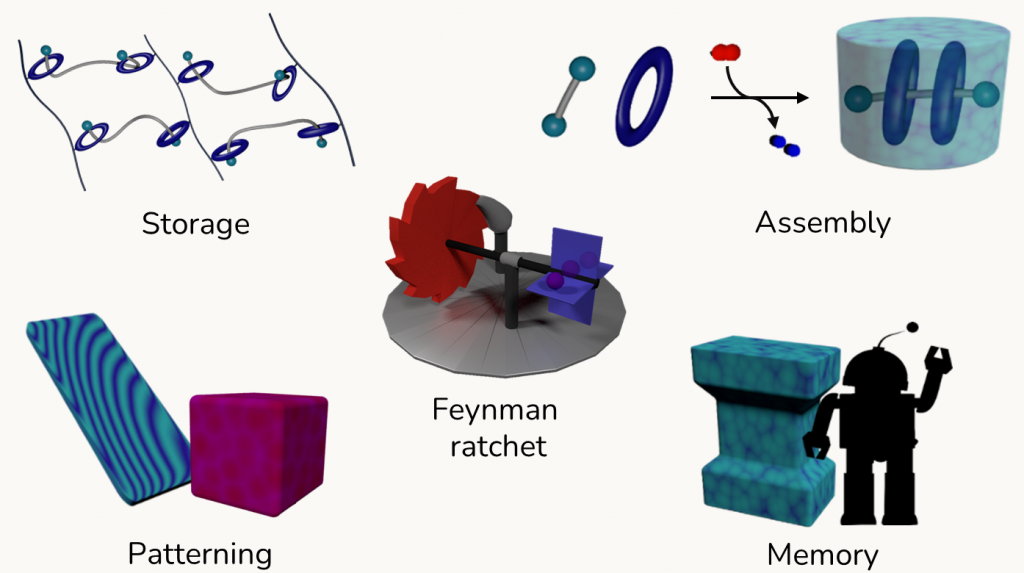
At the nanoscale, the mechanisms allowing to convert random – non-directional – energy into directed motion or work are so-called ratchet mechanisms. A core aspect of our science is engineering and exploiting molecular ratchets. These mechanisms have been popularized by Feynmann (here a famous video extract), and offered insights across disciplinary boundaries, e.g. in game theory, leading to the Parrondo paradox (here an interview on the topic). In this tutorial review we have described them in the context of chemistry (Angew. Chem. Int. Ed. 2023, e202309501).
Ratchets have been implemented into artificial molecular machines, allowing them to move in a preferential direction. Pioneers were Feringa and Leigh, who developed the first prototypes.
While molecular machines are at the forefront of nanotechnology, in most cases they operate in solution, affording directional motion: can we take advantage of the core mechanisms of molecular machines in other domains? Our goal is to broaden the application domain of ratchet mechanisms, to obtain energy-demanding processes also besides directional motion. With this approach, it could be possible to store energy in unconventional ways, develop mnemonic materials, synthetize thermodynamically unstable molecules, as well as other endergonic processes. On top of this, we aim at disclosing the relation between molecular ratchets and non-equilibrium phenomena such as oscillations and pattern formation, to reveal overarching principles governing non-equilibrium systems.
Practically speaking, in our group we design and synthesize molecules, study their behavior under energy inputs (chemical, electrical, photochemical), and rationalize the observed phenomena. Thus, we are placed at the crossroad between solution (organic, supramolecular) chemistry, soft matter science, and non-equilibrium physics.
Here you can hear Giulio discuss one of our research topics in the context of the Dream Chemistry Award 2021:
If you are interested in these themes, let’s get in touch!
If appropriate, consider also joining the group!
Funding
We are extremely grateful for all the funding that makes our research possible.







